A process under stress: why alkalinity becomes limiting
Nitrification naturally consumes alkalinity, about 7.14 mg as CaCO₃ per mg of ammonium oxidized and when the system can’t keep up, pH begins to fall. Even small shifts are enough to slow nitrifying bacteria and trigger instability. In practice, this shows up quickly in plants with soft influent water or those relying on ferric/alum for low P limits, both of which accelerate alkalinity depletion.
A low alkalinity environment doesn’t just weaken nitrification; it also increases the risk of nitrite accumulation and can elevate N₂O emissions, adding a climate impact dimension to what used to be considered a simple process-control issue.
The core challenges come down to:
Together, these factors make alkalinity control a critical lever for maintaining biological stability.
The case for calcium carbonate as a biological buffer
Many plants turn to caustic soda or hydrated lime to manage pH, but these chemicals often create more volatility than stability. Because they dissolve instantly, they can trigger rapid pH spikes and localized pH peaks. Additionally, they provide ongoing safety challenges for operators for their storage and dosing. More importantly, they only correct the symptom: low pH’, without improving the biological resilience of the system.
Calcium carbonate offers a fundamentally different approach. It dissolves only in the presence of acidity, releasing alkalinity gradually and exactly where it is needed. This self-regulating behavior stabilizes pH both at the tank level and within the micro-zones inside sludge flocs where nitrifiers live. At the same time, calcium ions strengthen the floc matrix by binding to negatively charged biopolymers, creating denser, more compact flocs that settle and dewater more effectively.
Because of this dual chemical and biological benefit, mineral alkalinity has become increasingly attractive to utilities seeking a more stable, predictable, and operator friendly way to support nitrification. Instead of sharp corrections and process swings, plants experience a smoother, more resilient biological environment that performs consistently under changing conditions.
Full-scale evidence from municipal operations
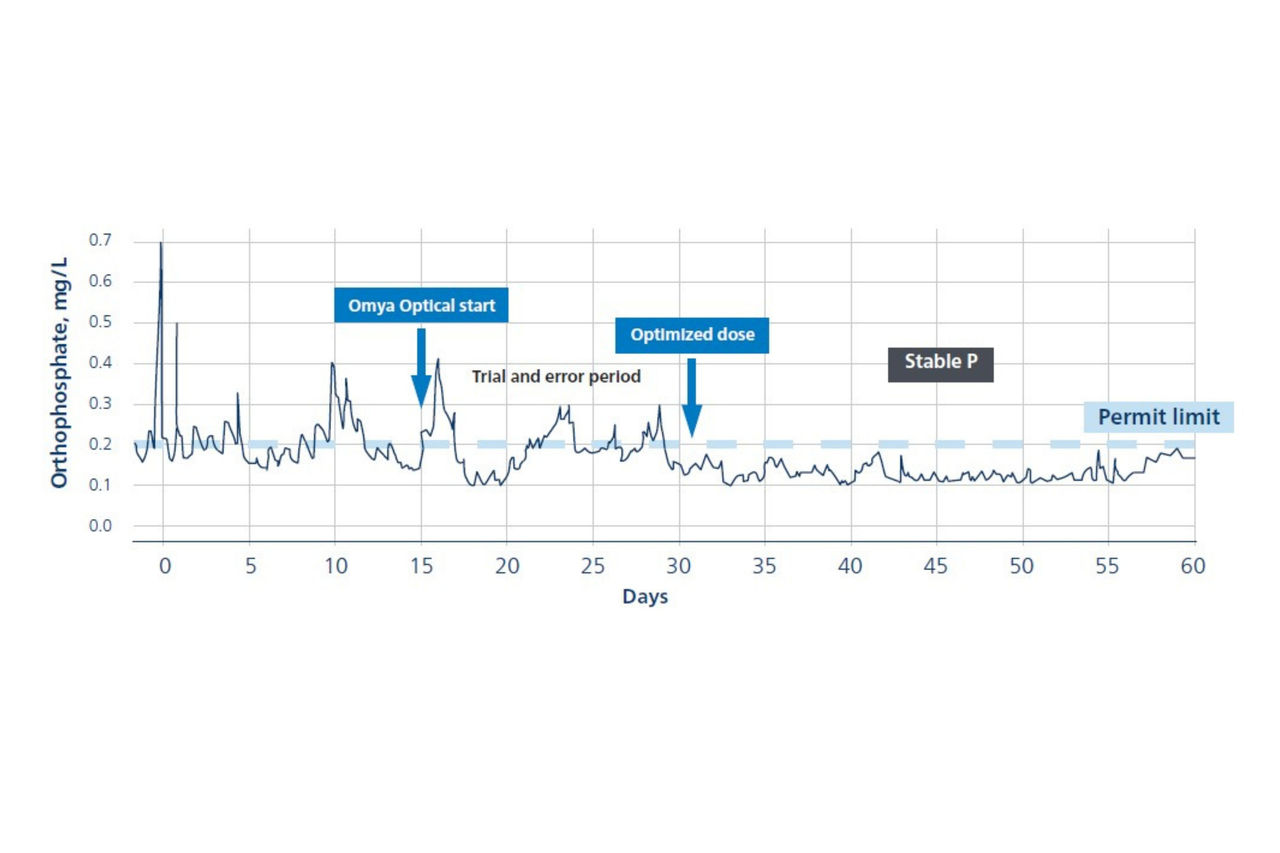
A recent two-month municipal trial shows how calcium carbonate-based buffering can improve biological stability, nutrient removal, and membrane performance in real world wastewater conditions. The facility operated membrane bioreactors with ferric-based phosphorus removal an approach that consumes alkalinity and depresses pH, especially when chemical dosing increases during periods of higher load. To avoid relying on caustic soda, operators introduced Omya Optical directly after primary clarification.
Once the dosing rate was optimized, the biological system immediately became more stable. pH remained within the target range of 7.0–8.0, and alkalinity consistently reached the plant’s preferred 40–60 mg/L as CaCO₃ range, with values up to 100 mg/L. This stable buffering environment supported both the activated sludge process and downstream membrane filtration.
Phosphorus removal performance strengthened as well. Orthophosphate concentrations stayed below the 0.2 mg/L permit limit throughout the optimized phase of the trial - something the plant struggled to achieve when alkalinity was low or dosing was inconsistent. Early underdosing made the contrast clear: compliance only stabilized once sufficient mineral buffering was established.
The membranes also responded positively. Calcium from Omya Optical bound with dissolved microbial substances, reducing organic fouling and improving permeability. This allowed the MBR system to operate more consistently, with fewer fluctuations in filtration performance.
To illustrate the performance shift, the data showed:
These improvements resulted not only from the alkalinity provided, but also from calcium’s role in improving floc structure and membrane behavior. By replacing caustic soda with a mineral-based alkalinity source, the utility strengthened biological stability, reduced operational risk, and supported reliable compliance- key advantages for municipal plants facing tighter nutrient limits and rising performance pressures.